Bubbles Protocol
Title: The Effect of the Composition of Water on Absorption of Carbon Dioxide
Topic Introduction- Ocean acidification is basically what happens when Co2 (Carbon dioxide) is chemically combined with water. The product of this combination is carbonic acid. This acid is what is causing the decay of the ocean's coral reefs and snail shells. Much of the water with carbonic acid present, also seem to affect the fish. As coral reefs are decaying, the diversity of the fish are lowering, and phytoplankton are becoming much less effective for the reefs.
Experimental Question: What are we trying to find out? How?
What is the effect of blowing carbon dioxide from our lungs into the bottles of water?
Pre-Lab Questions: Bubbles Protocol (answer in complete sentences)
1. What gas are you blowing into the water?
The gas that we are blowing into the water is Co2
2. What happens to the gas when you blow it into the water?
The gas is being absorbed by the water as we blow the gas in.
3. How are you measuring change in the water during this lab?
We are measuring change in the water by pH or the change of color.
4. What does measuring the pH of the water tell us?
Measuring the pH of the water, tell us whether the water is becoming more basic or acidic.
5. After studying the reactions above, how do you think carbonic acid will affect the pH of
salt water?
Seeing as "carbonic acid" has "acid" as part of its name, I predict that is will make the water more acidic.
Hypothesis: Bubbles Protocol- What did you predict would happen when you blow into the sea
water?
-If I blow carbon dioxide into the bottle of water, then the water should become more acidic.
Protocol: Briefly explain the protocol used in each of the parts of the experiment. What are the
dependent and independent variables? What are the constants?
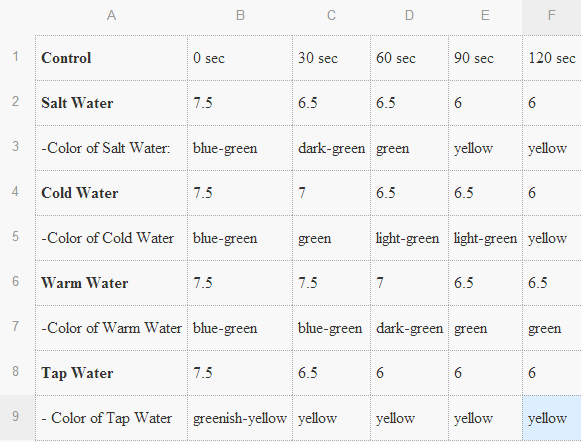
-This experiment required that we had one beaker, salt water, a universal indicator, and a straw. We placed 100ml
of water into the breaker. There were four different types of water that we used. These were salt, cold, tap,
and hot waters.We then placed a couple drops of the universal indicator into the water. To stop water from
plashing out, we placed plastic wrap over the beaker. The straw was pierced into the plastic wrap far enough
until it touched the water. Using our lungs we blew carbon dioxide into the water.
Dependent Variables: pH of the water over a 2 min period (measuring every 30 sec).
Independent Variables: The different types of water.
Data Analysis: (Post-Lab Questions)
1. As you blew through the straw, what were you adding to the water and how did that change the pH?
- When blowing into the water, I was adding carbon dioxide. Because carbon dioxide is acidic, it made the water have a lower pH.
2. What did the universal indicator tell us about the water?
- The universal indicator displayed the pH of the water through color.
3. What does this tell us about the effects of carbonic acid in ocean water?
- This tells us that the effects of carbonic in ocean water is dangerous to the life in the ocean as acid can make things decay
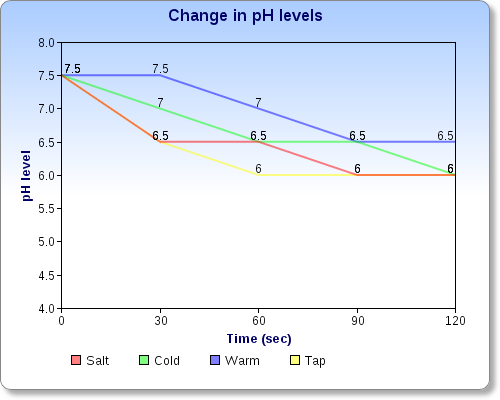
4. Based on the results of your experimental protocol, which factor affects the pH of the water most, temperature or salt?
- Based on our results, the salt water seems to change the pH of the water most.
Conclusion: Was your initial hypothesis correct? Why or why not? Explain. What did you learn about ocean acidification?
As I have said in my hypothesis, carbon dioxide did change the pH of the water.
The water absorbed my exhaled carbon dioxide while i blew into the straw. The water then formed carbonic acid. This is the product of the combination of carbon dioxide and water. This is also the cause of the loss of phytoplanton in the oceans because of it's acid property . As most of the types of water started at 7.5 pH, blowing the carbon dioxide into the water lowered the pH which makes it change into a more acidic state. The concluding pH ended up to usually become a 6.
Experimental Question: What are we trying to find out? How?
What is the effect of blowing carbon dioxide from our lungs into the bottles of water?
Pre-Lab Questions: Bubbles Protocol (answer in complete sentences)
1. What gas are you blowing into the water?
The gas that we are blowing into the water is Co2
2. What happens to the gas when you blow it into the water?
The gas is being absorbed by the water as we blow the gas in.
3. How are you measuring change in the water during this lab?
We are measuring change in the water by pH or the change of color.
4. What does measuring the pH of the water tell us?
Measuring the pH of the water, tell us whether the water is becoming more basic or acidic.
5. After studying the reactions above, how do you think carbonic acid will affect the pH of
salt water?
Seeing as "carbonic acid" has "acid" as part of its name, I predict that is will make the water more acidic.
Hypothesis: Bubbles Protocol- What did you predict would happen when you blow into the sea
water?
-If I blow carbon dioxide into the bottle of water, then the water should become more acidic.
Protocol: Briefly explain the protocol used in each of the parts of the experiment. What are the
dependent and independent variables? What are the constants?
-This experiment required that we had one beaker, salt water, a universal indicator, and a straw. We placed 100ml
of water into the breaker. There were four different types of water that we used. These were salt, cold, tap,
and hot waters.We then placed a couple drops of the universal indicator into the water. To stop water from
plashing out, we placed plastic wrap over the beaker. The straw was pierced into the plastic wrap far enough
until it touched the water. Using our lungs we blew carbon dioxide into the water.
Dependent Variables: pH of the water over a 2 min period (measuring every 30 sec).
Independent Variables: The different types of water.
Data Analysis: (Post-Lab Questions)
1. As you blew through the straw, what were you adding to the water and how did that change the pH?
- When blowing into the water, I was adding carbon dioxide. Because carbon dioxide is acidic, it made the water have a lower pH.
2. What did the universal indicator tell us about the water?
- The universal indicator displayed the pH of the water through color.
3. What does this tell us about the effects of carbonic acid in ocean water?
- This tells us that the effects of carbonic in ocean water is dangerous to the life in the ocean as acid can make things decay
4. Based on the results of your experimental protocol, which factor affects the pH of the water most, temperature or salt?
- Based on our results, the salt water seems to change the pH of the water most.
Conclusion: Was your initial hypothesis correct? Why or why not? Explain. What did you learn about ocean acidification?
As I have said in my hypothesis, carbon dioxide did change the pH of the water.
The water absorbed my exhaled carbon dioxide while i blew into the straw. The water then formed carbonic acid. This is the product of the combination of carbon dioxide and water. This is also the cause of the loss of phytoplanton in the oceans because of it's acid property . As most of the types of water started at 7.5 pH, blowing the carbon dioxide into the water lowered the pH which makes it change into a more acidic state. The concluding pH ended up to usually become a 6.
Shells Protocol
Title: The Dissolving of Shells Due to a High Rate of CO2
Topic Information- The rising acidity levels have a great effect on the many coral reefs and organism
shells of the ocean. Like coral reefs, the shells are made from calcium carbonate. Because of the rising
acid levels, these shells are slowly decaying over tame keeping the organism in a very vulnerable place.
The shells are used by the organisms as homes and protection against their predators.
Student Pre- lab Questions:
1. How do organisms make their shells? What are shells made of?
-The shells of the organisms are made by combing calcium and carbon together
making calcium carbonate.
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
I expect that a shell placed in an acidic solution will slowly decay over time.
3. What are sources of carbon dioxide and which of these sources are most likely to
affect ocean pH?
Much of the sources of carbon dioxide are from the many industrial factories. These
factories emit carbon dioxide from burning fossil fuel. Another is from the daily action
of just exhaling. These could both affect the oceans as carbon dioxide is everywhere
in the atmosphere
Hypothesis Shell Protocol: When placing an organism's shell into an acidic solution, then the shell
will decay over time.
Protocol:
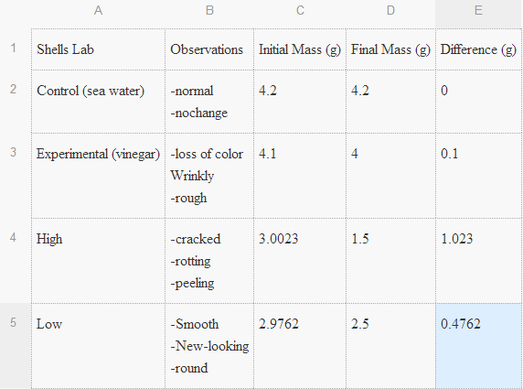
- We first measured the initial mass of two shells. Using two beakers, we placed 100
ml of water into one beaker and 100 ml of vinegar into another. We then placed the
shells both separately into the beakers. After a period of twenty minutes, we again
measured the mass of the snail shells (depending on which substance it was placed
in). In the end we recorded the differences between the before and after masses.
Dependent Variables: Mass of the shells
Independent Variables: Substances the shells were placed in
Constants: Beakers and amount of each substance.
Analysis/Discussion of Data
1. When you immersed the shells in vinegar how did you know that a reaction was
happening?
-You can tell that there was a reaction between the vinegar and the shells as many
air bubbles were surrounding the shell and then floating to the surface.
2. How did observing the shells in vinegar relate to how animals are affected by a
lower pH of ocean water?
- Observing the shells in vinegar relate to how animals are affected by lower pH
because seeing of how a low pH in the ocean can decay the environment,
many of the animals are dependent on the environments as homes and
resources.
3. How would shelled organisms be affected by a lower pH of ocean water?
Shelled organisms are affected by low pH levels because they rely on shells
as shelter from predators.
4. What are the primary functions of shell for these animals?
- The primary functions of a shell for the organisms are that they
use it for shelter and protection from predators.
5. Does it cost the animal energy to rebuild or repair their shell?
-Animal energy is required for repairing and rebuilding a shell.
Conclusion:
As my hypothesis said, shells are affected by a lower pH level and it was correct.
Watching the shell being placed in the acidic solution showed how damaging a low pH can be.
Over time the shell seemed to decay. When taken to get measured, the shell placed in the
experimental beaker got a drastic change in mass. Seeing this change shows fast the ocean
environment can change and many of the animals of the sea are dependent on their
normal environment.
shells of the ocean. Like coral reefs, the shells are made from calcium carbonate. Because of the rising
acid levels, these shells are slowly decaying over tame keeping the organism in a very vulnerable place.
The shells are used by the organisms as homes and protection against their predators.
Student Pre- lab Questions:
1. How do organisms make their shells? What are shells made of?
-The shells of the organisms are made by combing calcium and carbon together
making calcium carbonate.
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
I expect that a shell placed in an acidic solution will slowly decay over time.
3. What are sources of carbon dioxide and which of these sources are most likely to
affect ocean pH?
Much of the sources of carbon dioxide are from the many industrial factories. These
factories emit carbon dioxide from burning fossil fuel. Another is from the daily action
of just exhaling. These could both affect the oceans as carbon dioxide is everywhere
in the atmosphere
Hypothesis Shell Protocol: When placing an organism's shell into an acidic solution, then the shell
will decay over time.
Protocol:
- We first measured the initial mass of two shells. Using two beakers, we placed 100
ml of water into one beaker and 100 ml of vinegar into another. We then placed the
shells both separately into the beakers. After a period of twenty minutes, we again
measured the mass of the snail shells (depending on which substance it was placed
in). In the end we recorded the differences between the before and after masses.
Dependent Variables: Mass of the shells
Independent Variables: Substances the shells were placed in
Constants: Beakers and amount of each substance.
Analysis/Discussion of Data
1. When you immersed the shells in vinegar how did you know that a reaction was
happening?
-You can tell that there was a reaction between the vinegar and the shells as many
air bubbles were surrounding the shell and then floating to the surface.
2. How did observing the shells in vinegar relate to how animals are affected by a
lower pH of ocean water?
- Observing the shells in vinegar relate to how animals are affected by lower pH
because seeing of how a low pH in the ocean can decay the environment,
many of the animals are dependent on the environments as homes and
resources.
3. How would shelled organisms be affected by a lower pH of ocean water?
Shelled organisms are affected by low pH levels because they rely on shells
as shelter from predators.
4. What are the primary functions of shell for these animals?
- The primary functions of a shell for the organisms are that they
use it for shelter and protection from predators.
5. Does it cost the animal energy to rebuild or repair their shell?
-Animal energy is required for repairing and rebuilding a shell.
Conclusion:
As my hypothesis said, shells are affected by a lower pH level and it was correct.
Watching the shell being placed in the acidic solution showed how damaging a low pH can be.
Over time the shell seemed to decay. When taken to get measured, the shell placed in the
experimental beaker got a drastic change in mass. Seeing this change shows fast the ocean
environment can change and many of the animals of the sea are dependent on their
normal environment.