Virtual Lab #1
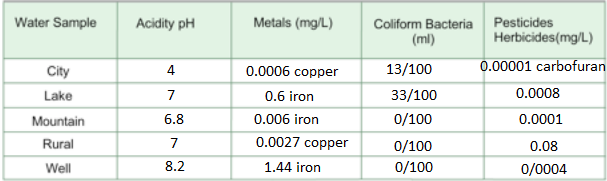
1: What contaminants were found in the surface water samples? What contaminants were found
in the groundwater samples?
Surface waters:
Groundwaters:
2: Why might groundwater and surface water have different contaminants?
These two bodies of water are placed in completely different locations allowing exposure to different kinds of contaminants.
3: Generally, farmers do not farm and industries do not build factories on the sides of mountains
in remote wilderness areas. These areas are usually not highly populated by people. What might
explain the high nitrate level in the mountain water in this activity?
The mountains probably contain natural nitrates which will run down the mountain slopes getting into the water.
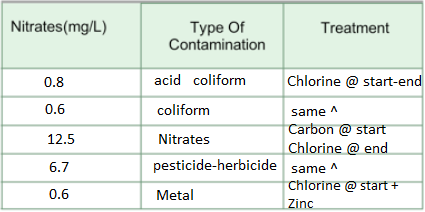
4: What is pH level, what are its characteristics and how does it contribute to pollution? What
chemicals are used in treating low pH levels?
pH levels determine how acidic or basic the water is. Chemicals determining in treating low pH
levels are determined by how far it is from the number 7. That number will be reflected and will
determine the number above 7 to see which chemicals are used.
5: Water in an old building, tested recently, showed high copper and iron content, and low pH
levels. A water reading taken 20 years before showed low pH levels, but only minimal traces of
copper and iron. If none of the new buildings on the same street showed signs of metallic
contaminants, but all reported lower than normal pH readings, how might these readings be
explained?
The pipes from the building containing concentrations of metals probably corroded. The new
buildings have new pipes that are not corroded so they don't contain any metals in the water.
in the groundwater samples?
Surface waters:
- Coliform
- Nitrates
- metals
Groundwaters:
- coliform
- acid
- pesticides
2: Why might groundwater and surface water have different contaminants?
These two bodies of water are placed in completely different locations allowing exposure to different kinds of contaminants.
3: Generally, farmers do not farm and industries do not build factories on the sides of mountains
in remote wilderness areas. These areas are usually not highly populated by people. What might
explain the high nitrate level in the mountain water in this activity?
The mountains probably contain natural nitrates which will run down the mountain slopes getting into the water.
4: What is pH level, what are its characteristics and how does it contribute to pollution? What
chemicals are used in treating low pH levels?
pH levels determine how acidic or basic the water is. Chemicals determining in treating low pH
levels are determined by how far it is from the number 7. That number will be reflected and will
determine the number above 7 to see which chemicals are used.
5: Water in an old building, tested recently, showed high copper and iron content, and low pH
levels. A water reading taken 20 years before showed low pH levels, but only minimal traces of
copper and iron. If none of the new buildings on the same street showed signs of metallic
contaminants, but all reported lower than normal pH readings, how might these readings be
explained?
The pipes from the building containing concentrations of metals probably corroded. The new
buildings have new pipes that are not corroded so they don't contain any metals in the water.