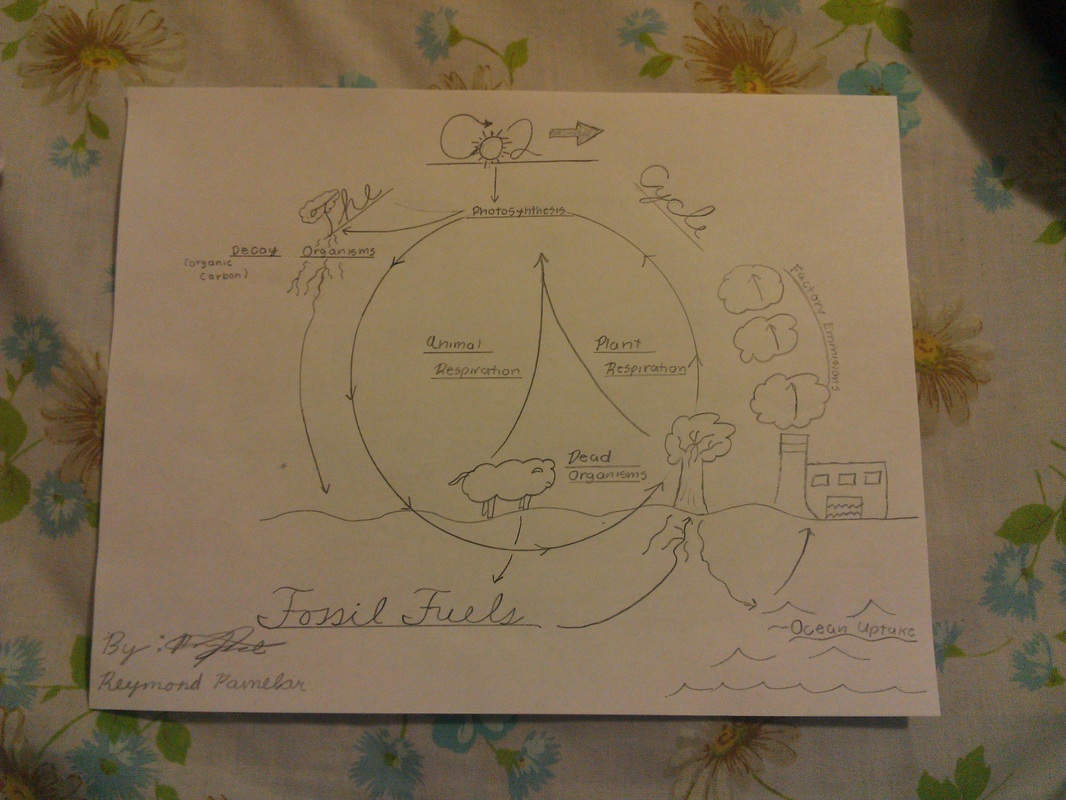
Name: Reymond Pamelar Date: 9/24/13
Cycling WebQuest
Background: In biogeochemical cycles (including carbon, water and nitrogen cycles), elements are
transported between the atmosphere, biosphere (living things), hydrosphere (water), and geosphere
(rocks, minerals, and soils). These cycles help us remember that Earth is a complex system.
Carbon Cycle:
Go to http://www.windows.ucar.edu/tour/link=/earth/Water/co2_cycle.html and answer these
questions:
1. Draw the carbon cycle (on a separate piece of paper)
Cycling WebQuest
Background: In biogeochemical cycles (including carbon, water and nitrogen cycles), elements are
transported between the atmosphere, biosphere (living things), hydrosphere (water), and geosphere
(rocks, minerals, and soils). These cycles help us remember that Earth is a complex system.
Carbon Cycle:
Go to http://www.windows.ucar.edu/tour/link=/earth/Water/co2_cycle.html and answer these
questions:
1. Draw the carbon cycle (on a separate piece of paper)
2. How does carbon exist in the atmosphere?
-Co2 is trapped in the atmosphere as gas and then builds up over time.
3. How are fossil fuels created? Explain.
-Fossil fuels are formed over millions of years from decomposing plants and animals.
4. Describe two ways that carbon enters the atmosphere.
1) Burning fossil fuels
2) The exhaling of living things which releases CO2
5. How are the oceans involved in the carbon cycle?
-Oceans soak up the carbon from the atmosphere.
6. How is the temperature of the Earth partly controlled by carbon?
-Carbon is what keeps the earth warm as the atmosphere traps it's heat.
7. What role do rocks have within the carbon cycle?
-Rocks are what the decayed plants and animals transfer their CO2.
Go to http://www.windows.ucar.edu/earth/climate/carbon_cycle.html to play the carbon cycle
game. You are a carbon atom!
8. Where are you starting within the carbon cycle?
-The Atmosphere
“Click to begin your journey”
9. How much of the atmosphere is made of carbon dioxide (CO2)?
-0.04% of the atmosphere is made of CO2
10. By how much has CO2 increased in the atmosphere during the past 150 years?
-It has increased by 30%
As you work through this game, take some notes about where you go as a carbon atom. Make sure
you visit all reservoirs!
-11. Next stop = Surface Ocean
What did you learn?
-The ocean takes up 90 gigatons of carbon a year which is more than what the land takes up.
12. Next stop = Deep Ocean
What did you learn?
-The deep ocean holds 65% of the earths carbon through circulation
The deep ocean accounts for more than 65 % of the Earth’s carbon.
How much carbon does the surface ocean absorb from the atmosphere each year?
-90 gigatons of carbon
True or False: When plants die and decay, they bring carbon into soil.
-True
13. Next stop = Marine Life
What did you learn?
-Marine life cannot survive without carbon but too much are bad for marine organisms like algae.
14. Next stop = Land Plants
What did you learn?
-Plants use photosynthesis to suck in carbon which help them grow fast.
15. Next stop = Soil
What did you learn?
-3% of earths carbon in held in soil which come from decomposed plants and animals.
When carbon enters the deep ocean, how long does it stay there?
-Hundreds of years
True or False: Phytoplankton are tiny plants and algae that float in the ocean and take up carbon dioxide.
-True
True or False: Plants both absorb CO2 from the atmosphere and release it into the atmosphere.
-True
Nitrogen Cycle:
Go to http://www.elmhurst.edu/~chm/onlcourse/chm110/outlines/nitrogencycle.html and
answer these questions.
16. What are the two conditions under which nitrogen will react with oxygen? (In other words, what is necessary for nitrogen in the air to combine with oxygen?)
-They will only react through high temperatures or through pressures found near lightning bolts.
17. What are the two compounds that are formed when nitrogen combines with oxygen?
-Nitric oxide and nitrogen oxide.
18. How does nitric acid (HNO3) form?
-When nitrogen oxide reacts with water.
19. Why is nitric acid (HNO3) important?
-Plants use it as a nutrient.
Go to: http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/N/NitrogenCycle.html and answer these questions.
20. What percentage of the air we breathe is nitrogen?
- It is 79% nitrogen.
21. Even though considerable nitrogen is available in the air, most plants do not use the nitrogen (N2) found in the air. Why not?
-The nitrogen have to be present in a fixed form.
22. In what compounds can plants use nitrogen?
- NO3, NH3, or (NH2)2CO
23. How do animals get the nitrogen they need?
-They get it from compounds of the plants.
24. Atmospheric nitrogen (N2) is pretty inert. This means that it does not easily break apart. When molecules do not break apart easily, it is difficult (or impossible) for organisms to use them as a nutrient source. As a result, nitrogen fixation is the term used to describe the process of breaking up N2.
a. What is atmospheric fixation?
- When the area around a lightning bolt lets atoms fuse with oxygen to create nitrogen oxides.
b. What is industrial fixation? [This is how artificial fertilizers are made.]
- When great temperatures and pressures combine atmospheric nitrogen and hydrogen to make amonia
c. What is biological fixation? (In your answer, describe the types of plants associated with the symbiotic relationship.)
To fix nitrogen through bacteria and archea.
Go to: http://www.physicalgeography.net/fundamentals/9s.html and answer these questions.
25. Draw the nitrogen cycle: On a separate piece of paper: (Remember there are other diagrams on the previous websites.) If you’re not sure what a term means, look through the reading and links for help.
26. Why is nitrogen needed by plants and animals?
-Plants use it for nutrients and animals need it for growth and healthiness.
Go to http://www.mbgnet.net/fresh/cycle/index.htm. Answer the following questions.
1. Define "water cycle".
- The water cycle is the different events of how water is purified and then recycled to the environment
2. What fraction of the Earth’s surface is covered in water?
-3/4's of the earth
3. What percentage of all the Earth’s water is in a form that is useable to humans and land animals?
-1% is useable
-Co2 is trapped in the atmosphere as gas and then builds up over time.
3. How are fossil fuels created? Explain.
-Fossil fuels are formed over millions of years from decomposing plants and animals.
4. Describe two ways that carbon enters the atmosphere.
1) Burning fossil fuels
2) The exhaling of living things which releases CO2
5. How are the oceans involved in the carbon cycle?
-Oceans soak up the carbon from the atmosphere.
6. How is the temperature of the Earth partly controlled by carbon?
-Carbon is what keeps the earth warm as the atmosphere traps it's heat.
7. What role do rocks have within the carbon cycle?
-Rocks are what the decayed plants and animals transfer their CO2.
Go to http://www.windows.ucar.edu/earth/climate/carbon_cycle.html to play the carbon cycle
game. You are a carbon atom!
8. Where are you starting within the carbon cycle?
-The Atmosphere
“Click to begin your journey”
9. How much of the atmosphere is made of carbon dioxide (CO2)?
-0.04% of the atmosphere is made of CO2
10. By how much has CO2 increased in the atmosphere during the past 150 years?
-It has increased by 30%
As you work through this game, take some notes about where you go as a carbon atom. Make sure
you visit all reservoirs!
-11. Next stop = Surface Ocean
What did you learn?
-The ocean takes up 90 gigatons of carbon a year which is more than what the land takes up.
12. Next stop = Deep Ocean
What did you learn?
-The deep ocean holds 65% of the earths carbon through circulation
The deep ocean accounts for more than 65 % of the Earth’s carbon.
How much carbon does the surface ocean absorb from the atmosphere each year?
-90 gigatons of carbon
True or False: When plants die and decay, they bring carbon into soil.
-True
13. Next stop = Marine Life
What did you learn?
-Marine life cannot survive without carbon but too much are bad for marine organisms like algae.
14. Next stop = Land Plants
What did you learn?
-Plants use photosynthesis to suck in carbon which help them grow fast.
15. Next stop = Soil
What did you learn?
-3% of earths carbon in held in soil which come from decomposed plants and animals.
When carbon enters the deep ocean, how long does it stay there?
-Hundreds of years
True or False: Phytoplankton are tiny plants and algae that float in the ocean and take up carbon dioxide.
-True
True or False: Plants both absorb CO2 from the atmosphere and release it into the atmosphere.
-True
Nitrogen Cycle:
Go to http://www.elmhurst.edu/~chm/onlcourse/chm110/outlines/nitrogencycle.html and
answer these questions.
16. What are the two conditions under which nitrogen will react with oxygen? (In other words, what is necessary for nitrogen in the air to combine with oxygen?)
-They will only react through high temperatures or through pressures found near lightning bolts.
17. What are the two compounds that are formed when nitrogen combines with oxygen?
-Nitric oxide and nitrogen oxide.
18. How does nitric acid (HNO3) form?
-When nitrogen oxide reacts with water.
19. Why is nitric acid (HNO3) important?
-Plants use it as a nutrient.
Go to: http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/N/NitrogenCycle.html and answer these questions.
20. What percentage of the air we breathe is nitrogen?
- It is 79% nitrogen.
21. Even though considerable nitrogen is available in the air, most plants do not use the nitrogen (N2) found in the air. Why not?
-The nitrogen have to be present in a fixed form.
22. In what compounds can plants use nitrogen?
- NO3, NH3, or (NH2)2CO
23. How do animals get the nitrogen they need?
-They get it from compounds of the plants.
24. Atmospheric nitrogen (N2) is pretty inert. This means that it does not easily break apart. When molecules do not break apart easily, it is difficult (or impossible) for organisms to use them as a nutrient source. As a result, nitrogen fixation is the term used to describe the process of breaking up N2.
a. What is atmospheric fixation?
- When the area around a lightning bolt lets atoms fuse with oxygen to create nitrogen oxides.
b. What is industrial fixation? [This is how artificial fertilizers are made.]
- When great temperatures and pressures combine atmospheric nitrogen and hydrogen to make amonia
c. What is biological fixation? (In your answer, describe the types of plants associated with the symbiotic relationship.)
To fix nitrogen through bacteria and archea.
Go to: http://www.physicalgeography.net/fundamentals/9s.html and answer these questions.
25. Draw the nitrogen cycle: On a separate piece of paper: (Remember there are other diagrams on the previous websites.) If you’re not sure what a term means, look through the reading and links for help.
26. Why is nitrogen needed by plants and animals?
-Plants use it for nutrients and animals need it for growth and healthiness.
Go to http://www.mbgnet.net/fresh/cycle/index.htm. Answer the following questions.
1. Define "water cycle".
- The water cycle is the different events of how water is purified and then recycled to the environment
2. What fraction of the Earth’s surface is covered in water?
-3/4's of the earth
3. What percentage of all the Earth’s water is in a form that is useable to humans and land animals?
-1% is useable